By Joanne Thiessen Martens
Ph.D. Candidate, Department of Soil Science, University of Manitoba
Organic farming has a phosphorus problem. Exporting nutrients off the farm without replacing them depletes soil phosphorus (P) and eventually reduces crop yields. This problem is especially common in long-term organic systems if livestock manure is scarce and the soil is alkaline.
Meanwhile, our lakes have a different phosphorus problem. Nutrients exported from farms in agricultural products travel through the food chain (including our bodies), enter waste treatment systems and finally waterways, where they can contaminate water and stimulate algal blooms.
What if it were possible to solve both problems at once, by recycling ‘waste’ nutrients back to organic farmland in a clean, easy-to-use, affordable, phosphorus-rich fertilizer? According to Kim Wilton, an organic grain farmer in central Manitoba, that possibility “would be a game-changer” for her operation.
That possibility is in the process of becoming a reality in Canada.
The phosphorus problem #
The phosphorus problem is more critical in certain areas. On one organic grain and livestock operation in Manitoba, the lack of a suitable phosphorus source is the farm’s greatest challenge, despite having a sizeable livestock herd that is grazed rotationally and provides manure for compost. Neighbouring farms don’t have excess manure to sell, and other permitted phosphorous sources (e.g., rock phosphate) are either ineffective in the alkaline, clay soils or too expensive to apply on a field scale (e.g., bonemeal).
Crops, particularly legumes, have stunted growth, the farmer notes. “That severely affects our ability to grow a good green manure crop to replenish nutrients. We need a solution to this problem soon or it threatens our future in organic production.”
Struvite: A recycled fertilizer #
Recycling nutrients from human waste back to farmland could address the phosphorus deficiency and the degradation of water bodies. For example, in this ‘circular economy,’ struvite can be recovered from municipal wastewater and repurposed as phosphorus fertilizer. Struvite is a mineral consisting of magnesium, phosphate, ammonium, and water held together in a crystalline structure that precipitates naturally under the right conditions. When produced under carefully controlled conditions in wastewater treatment facilities, struvite crystals contain very low levels of contaminants and form granules that can be used as a fertilizer.
Struvite is unique among fertilizers. Unlike common synthetic fertilizers, struvite doesn’t dissolve well in water. This gives it a reputation as a ‘slow-release’ fertilizer that does not overwhelm the soil system with a large pulse of nutrients. However, struvite is more soluble in soil than rock phosphate, especially in alkaline soils where rock phosphate provides little benefit to crops. With an N-P-K analysis of 5-28-0, struvite is a more concentrated phosphorus source than most fertilizers permitted in organic production, with a lower price per unit of phosphorus. Its low solubility may reduce the risk of losses to the environment by runoff.
These qualities have led many experts to recommend that struvite be permitted for use in organic agriculture to help address phosphorus deficiencies. Many organic farmers agree. Dan DeRuyck, an organic grain and beef producer in south-central Manitoba, welcomes a new way to replenish soil phosphorus on his farm.
“I am hoping that struvite will be a product we can use in organic production,” DeRuyck says. “I like that it comes from a recycled process instead of being mined, which we will eventually run out of. This product will give me another tool in my crop production.”
Struvite from livestock- or plant-based sources was added to the 2020 Canadian Organic Standards Permitted Substances List (PSL). However, even though the principles of organic agriculture emphasize recycling of materials, human-sourced struvite is not permitted due to concerns about contaminants. Unfortunately, the only commercially available struvite in Western Canada is human-sourced.

Managing a novel nutrient source #
Because of struvite’s unique properties, we know very little about how to manage it well as a fertilizer. Research has shown that soil properties, plant species, and struvite granule size can all influence crop response. Also, phosphorus may continue to be released long after the growing season, even in the second or third year after application.
With the support of Organic Science Cluster (OSC) 3, I conducted field experiments in 2017–2019 as part of my doctoral research to test the effect of different struvite application rates on the yields of spring wheat, flax, and alfalfa-grass forage. The study site near Libau, Manitoba, was typical of many organic farms in the region. With a history of alfalfa hay export and no application of nutrient sources, soil test phosphorus was extremely low and crop productivity had declined over the years. The alkaline soil provided a challenging testing ground for struvite, which tends to work better in neutral or acidic soils.
For wheat and flax, we applied struvite in the furrow with the seed at three different rates, as well as an unfertilized control treatment. The experiment was repeated in each of the three study years. For the alfalfa-grass forage, we applied struvite to an existing forage stand in spring of 2017 by banding it about an inch below the surface, again at three different rates plus a control. We monitored the crop response over a three-year period.
Results were mixed. Wheat showed a moderate response, with slightly higher yields attained with every increase in struvite application rate. The highest application rate, which was double the recommended rate for that soil, yielded an average of 38.9 bu/ac over the three years of the study, a 35 percent increase over the unfertilized control (Table 1). Flax yields, on the other hand, did not change with struvite application, hovering around 20 bu/ac for all treatments.
Table 1. Wheat and flax grain yields when fertilized with different rates of struvite in experiments. Results are averaged over three study years.
| Phosphorus* application rate, as struvite | Wheat yield (bu/ac) | Flax yield (bu/ac) |
| 0 lb/ac P | 28.5 | 19.5 |
| 18 lb/ac P | 31.7 | 19.7 |
| 27 lb/ac P | 34.6 | 19.4 |
| 36 lb/ac P | 38.9 | 20.3 |
*To convert the P application rate to P2O5, multiply by 2.3.
The alfalfa-grass forage responded strongly to struvite, especially in the second and third years after application. In the first year (2017), the highest struvite application rate increased forage yield by 65 percent over the unfertilized control treatment (Figure 1). In the second year, the plots receiving the highest struvite rate yielded more than double the control; in the third year, the difference was more than triple. This pattern was caused partly by increases in yield over time in the plots with the high application rate, but also by a decline in the unfertilized control yields over time.
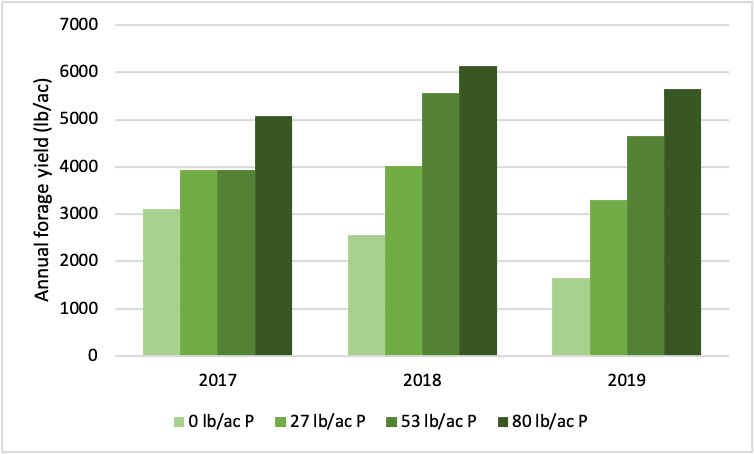
Figure 1. Total annual forage yield (sum of two cuts) from alfalfa-grass forage fertilized with different application rates of phosphorus as struvite. Struvite was applied in the spring of 2017.
Dr. Kim Schneider, assistant professor at the University of Guelph and co-lead of this
OSC3 project, finds the difference in response among crops to be particularly interesting.
“We need to continue to understand the mechanism by which some crops can access struvite and some can’t, and then put this to work in our farming systems!” Dr. Schneider explains.
One approach is to use the slow-release properties strategically in our crop rotations. In the wheat and flax experiments, we found remnants of struvite granules in the soil one year after application, meaning that more struvite could be released for the next crop. By better understanding how struvite interacts with soil over time and how different crops use struvite, we may be able to develop specific guidelines for when to apply struvite in a crop rotation to provide the most benefit to a series of crops.
Another question was raised: how will struvite’s slow-release over time affect the potential loss to the environment?
Dr. Henry Wilson, research scientist at the Brandon Research and Development Centre of Agriculture and Agri-Food Canada and the other co-lead of this project, is addressing this. By applying struvite to one of two small watersheds within an alfalfa-grass hay field and monitoring the phosphorus in the spring snowmelt runoff, it’s possible to see whether phosphorus from struvite is leaking out of the soil into the environment.
The preliminary results are very promising. Soil test phosphorus has increased in the part of the field fertilized with struvite but phosphorus concentration in the snowmelt runoff is the same as the area where no struvite was applied. Wilson explains, “this indicates that there doesn’t seem to be a large amount of that residual soil phosphorus being lost through leaching into snowmelt.”

Future outlook #
Our results so far suggest that struvite can be a very good fit as a soil amendment in organic cropping systems. It is an affordable phosphorus input that is effective in alkaline soils for at least some crops, with low risk of environmental losses. It also aligns well with organic principles. There is still much to learn about how to use it most effectively, but our knowledge is growing with every new experiment.
The main hurdle is to either develop livestock- or plant-sourced struvite supplies or to add human-sourced struvite to the PSL.
“If it gains full certification, struvite represents a viable option for organic farmers to help replenish phosphorus reserves on their farms, something particularly relevant to Prairie regions without a supply of livestock manure,” says Schneider.
That would be a game-changer indeed.
The Organic Science Cluster 3 is led by the Organic Federation of Canada in collaboration with the Organic Agriculture Centre of Canada at Dalhousie University, and is supported by the AgriScience Program under Agriculture and Agri-Food Canada’s Canadian Agricultural Partnership (an investment by federal, provincial and territorial governments) and over 70 partners from the agricultural community.
This magazine may be cited as: Geldart, E.. Graves, M.E., Boudreau, N., Wallace, J., and Hammermeister, A.M. (Editors). 2022. Organic Science Canada. Volume 4. Organic Federation of Canada, Montreal, QC and Dalhousie University, Truro, NS. 40 pp. www.dal.ca/oacc/oscIII



